 Counterfeiting, theft and criminal damage are continuous challenges faced while pharmaceutical packaging by companies around the globe. With the stakes so high for companies, the importance of the packaging cannot be understated.
Counterfeiting, theft and criminal damage are continuous challenges faced while pharmaceutical packaging by companies around the globe. With the stakes so high for companies, the importance of the packaging cannot be understated.
A compromised pharmaceutical product carries many risks. Most importantly, for the patient who may take medicines which have been tampered with or are counterfeit – posing potential serious consequences for their health. Reputational damage and commercial issues for the manufacturer or supplier of the medicine that has been copied or tampered with are also important considerations to take into account.
Counterfeit Products
The World Health Organisation (WHO) has estimated that somewhere close to 10% of all medicines sold worldwide are counterfeit. This claim is supported by another startling statistic from the European Consumers’ Organisation report that indicates a 400% increase in the number of counterfeit medicines seized by officials at EU borders.
Tackling issues such as counterfeiting and breached products alongside regulatory demands concerning contamination and storage, it is no great surprise that the pharmaceutical industry invests heavily in packaging.
Key issues for packaging in the sector are:
- Protection from airborne contamination
- Preventing moisture, gas or light damage
- Security
Folding Boxboard (FBB) is used extensively in pharmaceutical packaging; its robust high-strength grammage being robust enough to limit damage to packaging during the transportation of items. It also successfully prevents moisture, gas and light damage.
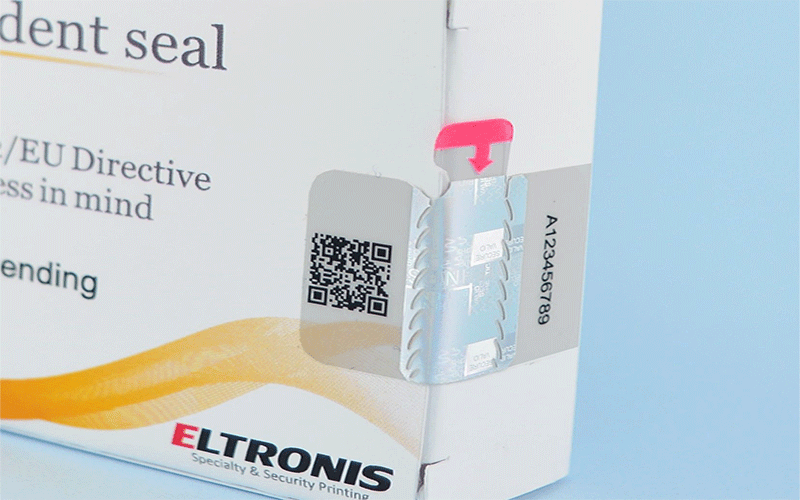
As well as providing protection, packaging must also be tamper-evident. Having tamper-evident packaging ensures that a stockist, patient or medic can easily tell if the medicine has potentially been compromised. If you had received a damaged or opened box of prescription antibiotics, would you be comfortable consuming the contents?

Tamper – Evident Packaging
Tamper-evident packaging which is also known and referred to as (TEP) offers brands and businesses a variety of benefits, fundamentally security.
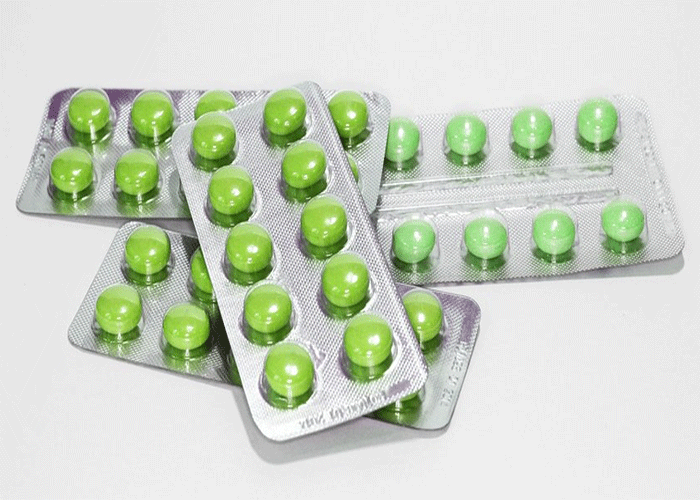
Alongside tamper-evident packaging sits tamper-resistant packaging; each satisfying a different requirement for the healthcare industry. In the United States, the FDA describes tamper-evident packaging as having more than one visible indicator of the product being compromised. Clear blister packages are sealed with foil, for an added layer of protection. If the blister package had been compromised, an indicator would be the damage to the foil seal. This simple, yet effective use of packaging ensures that we are further able to keep consumers and pharmaceutical products safe. Another added benefit is that the packaging often contains a warning message, alerting the user of this type of tampering.
Other Types of Tamper – Evident Packaging:
- A tamper-evident shrink band is an excellent option for products that have a lid or caps. An example of these would be an ice-cream lid or food jar lids.
- Tamper-evident tapes are a fantastic choice to add security to boxes, envelopes, pallets, crates, and bags.
Tamper-evident packaging and labeling are vital to providing security. The FDA has issued rulings around the standardization for pharmaceutical labeling and product details since 1906.
Today, pharmaceutical companies invest heavily in R&D for packaging – to produce the physical product itself from damage, to protect their intellectual property, and ultimately to protect the health of the patient consuming the medicine.
Packaging developments in the sector have included the addition of active and intelligent technologies such as; unique ID codes to boxes or containers, colour-changing inks that indicate exposure to extreme heat or cold, RFID tags (radio frequency identification) that allow the ‘track and trace’ of medication, and the use of printed electronic features on the packaging.
It is often said that the only constant is change – this is unquestionably true for the packaging manufacturers working alongside the pharmaceutical sector, as they strive to offer attractive and safe medicines around the globe.